Are your slides sticky enough for tricky tissues?
Objectives
Evaluate and compare the tissue adhesion performance of slide brands across different tissue types in a common IHC/ISH platform.
Evaluate and compare the tissue adhesion performance of slide brands across different tissue types in a common IHC/ISH platform.
Background
Adhesion slides are widely preferred for IHC to aid in securingtissue sections to the slide and prevent reworks that could potentially postpone a patient diagnosis and drive-up costs in the lab. The cost of reworking a failed IHC slide due to poor tissue adhesion is estimated to be ~$80 per slide, considering the reagent cost and workload administration.1 Adhesion slides reinforce tissue adherence and integrity, minimizing the need to recut and restain the sample to ensure proper tissue morphological characteristics.
Adhesion slides are widely preferred for IHC to aid in securingtissue sections to the slide and prevent reworks that could potentially postpone a patient diagnosis and drive-up costs in the lab. The cost of reworking a failed IHC slide due to poor tissue adhesion is estimated to be ~$80 per slide, considering the reagent cost and workload administration.1 Adhesion slides reinforce tissue adherence and integrity, minimizing the need to recut and restain the sample to ensure proper tissue morphological characteristics.
Materials and Methods
Tissue adhesion plays a crucial role in Immunohistochemical (IHC) staining due to the aggressive nature of the antigen retrieval process. To evaluate adhesion, IHC staining was performed on a Ventana BenchMark ULTRA using three different tissue types: tonsil (1 tissue section), skin (2 different tissue sections), and breast (3 different tissue sections). These tissues were selected based on their variable adhesion challenges, with expectations of tissue wash or detachment based on histological expertise.2 Tonsil sections very rarely become detached, where breast sections are well known to have tissue wash. Due to the fact that over 300,000 Americans alone are diagnosed with invasive breast cancer each year, half of the testing focus was on breast tissue.3 All tissues sections were cut at 4 microns and dried at 60°C for 30 minutes in a StatLab High Capacity Slide Dryer. As a stress test, an aggressive antigen retrieval method, CC1 standard at 95°C, was used for all samples, and antibody incubation was 32 minutes at 36°C. After staining, each tissue section was graded (see grading scale A) microscopically for tissue adhesion (see Table 1). Slides which scored a 3 or lower, indicating that 50% or less of the tissue remained on the slide, were considered to be a “Failure” – indicating that the slide would need to be recut and restained to adequately assess the tissue sample.
- StatLab KT5+™ Premium Adhesion Slides
- Matsunami TOMO® Adhesion Slides
- Epredia SuperFrost™ Plus Adhesion Slides
- Tissue: Skin Shaves, Skin, Tonsil, Benign Breast, Breast
Cancer A, Breast Cancer B - Ventana BenchMark ULTRA IHC Stainer
- StatLab High Capacity Slide Dryer
Tissue adhesion plays a crucial role in Immunohistochemical (IHC) staining due to the aggressive nature of the antigen retrieval process. To evaluate adhesion, IHC staining was performed on a Ventana BenchMark ULTRA using three different tissue types: tonsil (1 tissue section), skin (2 different tissue sections), and breast (3 different tissue sections). These tissues were selected based on their variable adhesion challenges, with expectations of tissue wash or detachment based on histological expertise.2 Tonsil sections very rarely become detached, where breast sections are well known to have tissue wash. Due to the fact that over 300,000 Americans alone are diagnosed with invasive breast cancer each year, half of the testing focus was on breast tissue.3 All tissues sections were cut at 4 microns and dried at 60°C for 30 minutes in a StatLab High Capacity Slide Dryer. As a stress test, an aggressive antigen retrieval method, CC1 standard at 95°C, was used for all samples, and antibody incubation was 32 minutes at 36°C. After staining, each tissue section was graded (see grading scale A) microscopically for tissue adhesion (see Table 1). Slides which scored a 3 or lower, indicating that 50% or less of the tissue remained on the slide, were considered to be a “Failure” – indicating that the slide would need to be recut and restained to adequately assess the tissue sample.
Table 1.
Tissue adhesion scoring results after aggressive antigen retrieval.ssess the tissue sample.
Tissue adhesion scoring results after aggressive antigen retrieval.ssess the tissue sample.
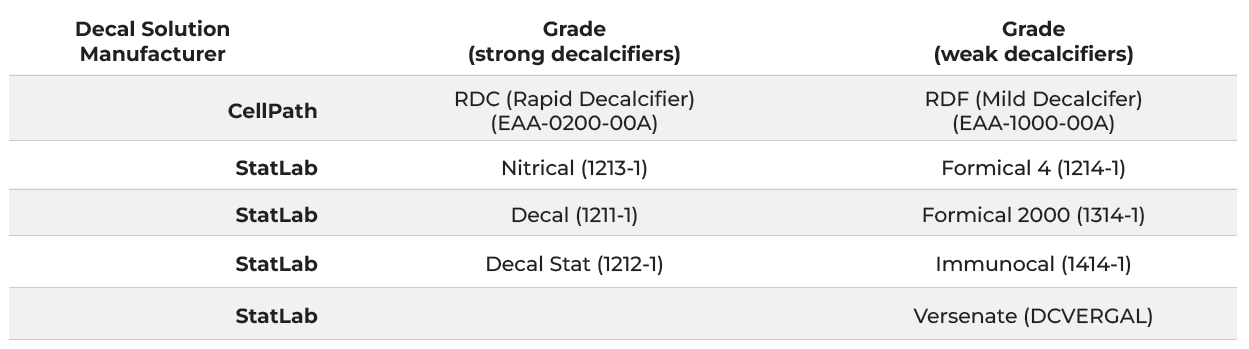
Table 2.
Failure rates by brand.
Failure rates by brand.
Grading Scale A.
Adhesion Scoring Criteria: % of tissue intact post-staining.
Adhesion Scoring Criteria: % of tissue intact post-staining.
Failure defined as any slide scored at a 1, 2, or 3 out of 5 for tissue adhesion, indicating that 50% or less of the tissue remained on the slide after staining.
Illustration 1.
Breast tissue after processing and staining.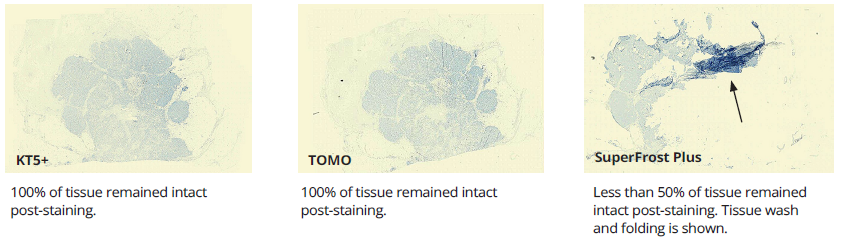
Breast tissue after processing and staining.

Illustration 2.
Skin shaves after processing and staining.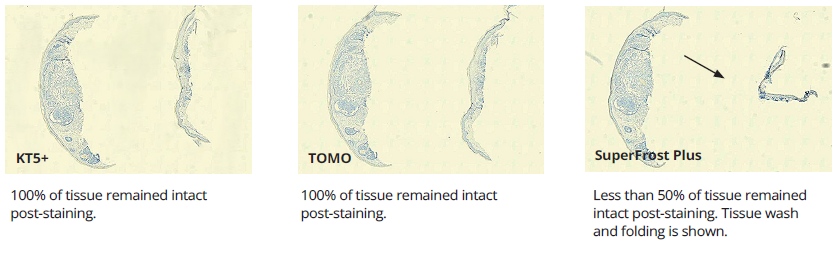
Skin shaves after processing and staining.

Results
Performance variation in adhesion slides was apparent in IHC testing. Tonsil, widely considered to be a low-difficulty tissue, performed well with minimal failures. Variations in adhesion performance became more noticeable with medium-difficulty tissues like skin but was most substantial with high-difficulty tissues like breast. Failure rates due to tissue wash, folding, and separation were observed at medium-high rates of failure in SuperFrost+ and TOMO slides. Failure rates with KT5+ were the lowest recorded. (see Table 2).
Performance variation in adhesion slides was apparent in IHC testing. Tonsil, widely considered to be a low-difficulty tissue, performed well with minimal failures. Variations in adhesion performance became more noticeable with medium-difficulty tissues like skin but was most substantial with high-difficulty tissues like breast. Failure rates due to tissue wash, folding, and separation were observed at medium-high rates of failure in SuperFrost+ and TOMO slides. Failure rates with KT5+ were the lowest recorded. (see Table 2).
Conclusion
After testing of slide adhesion across different types of slides, this study shows that not all adhesion slides are created equal. This study does suggest to labs that it is important to determine what the needs are for the laboratory based on tissue types and protocols used. The results show that KT5+ slides exhibit high tissue adhesion with even the most aggressive antigen retrieval procedures and maintain tissue integrity with more difficult specimens.
After testing of slide adhesion across different types of slides, this study shows that not all adhesion slides are created equal. This study does suggest to labs that it is important to determine what the needs are for the laboratory based on tissue types and protocols used. The results show that KT5+ slides exhibit high tissue adhesion with even the most aggressive antigen retrieval procedures and maintain tissue integrity with more difficult specimens.
REFERENCES
1. https://elearn.nsh.org/products/slide-surface-chemistry-understanding-an-essential-link-to-obtaining-quality-ihc-staining-results
2. https://www.leicabiosystems.com/us/knowledge-pathway/an-introduction-to-specimen-processing/
3. www.cancer.org/cancer/types/breast-cancer/understanding-a-breast-cancer-diagnosis/breast-cancer-her2-status.html3.
4. https://www.biosb.com/wp-content/uploads/Final-Hydrophilic-Plus-Slides-for-Molecular-Pathology.pdf
1. https://elearn.nsh.org/products/slide-surface-chemistry-understanding-an-essential-link-to-obtaining-quality-ihc-staining-results
2. https://www.leicabiosystems.com/us/knowledge-pathway/an-introduction-to-specimen-processing/
3. www.cancer.org/cancer/types/breast-cancer/understanding-a-breast-cancer-diagnosis/breast-cancer-her2-status.html3.
4. https://www.biosb.com/wp-content/uploads/Final-Hydrophilic-Plus-Slides-for-Molecular-Pathology.pdf
The findings and conclusions presented are based on internal research conducted by StatLab Medical Products under specific testing conditions. These results should be considered preliminary and are not intended to replace independent external studies or peer-reviewed research. Performance may vary depending on laboratory protocols, environmental factors, and specimen characteristics.StatLab Medical Products makes no warranties, express or implied, regarding the completeness, accuracy, or reliability of the information provided. The mention of third-party products is for comparative purposes only and does not imply endorsement or criticism. Readers are encouraged to conduct their own evaluations and consult appropriate regulatory and scientific sources before making purchasing or clinical decisions.StatLab Medical Products shall not be held liable for any direct, indirect, incidental, or consequential damages arising from the use or reliance on the information contained in this document.
Recent Posts
-
Amyloid Sections Washing Off? Let’s get to the bottom of this tricky stain.
Have you ever wondered how an 8–10 µm amyloid section washes off with aggressive alkaline rinses of
-
Thinking of going xylene-free? Here’s what labs should know.
Histotechs know the dangers and health risks associated with xylene, and interest in greener labs co
-
Manual vs. Semi-Auto Microtomes: Why the small difference makes a big impact.
Manual microtomes have long been a reliable choice in histology labs, but semi-automatic models, lik





